With molar mass periodic table4/18/2024  Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Magnesium (Mg), Group 2, Atomic Number 12, s-block, Mass 24.305. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. Multiply the number of each atom by its atomic weight (found on the Periodic Table of the Elements). History textPodcasts The Naked Scientists. Water is made up of 2 hydrogens and 1 oxygen.Ģ. The molar mass is calculated by taking the sum of the atomic weights of all the atoms which form the molecule.įirst determine the number of each atom in the formula.ġ. When there is parenthesis's around a group of atoms we must multiply each atom by the subscript to get the total number of each element.Ĭa(NO 3) 2 1 Calcium 2 Nitrogen and 6 Oxygen -> 3 x 2 Reading Compounds that have (parenthesis's). One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. If there is only 1 atom we do not use subscripts. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. The subscript belongs to the element it follows and indicates how many of that atom there are. 
Reading Simple Chemical Formulas- Each element begins with a CAPITAL letter. The units are atomic mass units ( amu ).ġamu =1.660 538 782×10 –27 kg 1 amu is 1/12 the mass of a carbon-12 atom which has a mass of 12.0000. 6.1: Early History of the Periodic Table. In this chapter we will learn about the connection between the two, and how the electronic structure is related to the macroscopic properties of the elements. The original periodic table emphasized the reactivity of the elements. Molecular Mass is the mass of a given molecule (NOT MOLES of molecules). The modern periodic table emphasizes the electronic structure of atoms. Molecular Weight is the molar mass of a COVALENT compound. The unit for molar mass (note it is the mass of a mole) is grams/mole.Ītomic Weight is the molar mass of an element.įormula Weight is the molar mass of an IONIC compound. 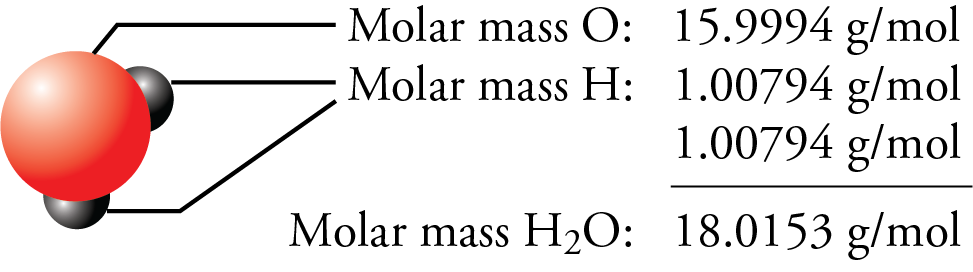
Visualize trends, 3D orbitals, isotopes, and mix compounds. Interactive periodic table showing names, electrons, and oxidation states. 
Molar Mass is the mass of one mole of a substance (6.02 x 10 23 formula units). For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. (Molar Mass, Molecular Weight, Gram Formula Mass) 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
